Liver Cancer Diagnostics Market Forecast 2026–2036: Market to Reach USD 25.8 Billion by 2036 at 8.5% CAGR
DE, UNITED STATES, March 20, 2026 /EINPresswire.com/ --
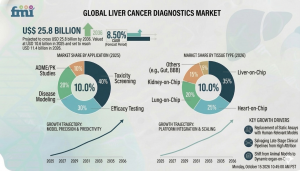
The global Liver Cancer Diagnostics Market is valued at USD 11.4 billion in 2026 and is projected to reach USD 25.8 billion by 2036, expanding at a CAGR of 8.5%. Having stood at USD 10.6 billion in 2025, the market reflects a strong growth trajectory driven by precision diagnostics, AI integration, and increasing global cancer screening demand.
The Liver Cancer Diagnostics Market is gaining strategic importance as healthcare systems prioritize early detection, staging accuracy, and non-invasive monitoring. Rising hepatitis prevalence and advancements in biomarker discovery are reshaping diagnostic pathways, making liver cancer detection faster, more accurate, and more scalable.
Market snapshot: global market 2026 - 2036
Market size 2026: USD 11.4 billion
Market size 2036: USD 25.8 billion
CAGR (2026–2036): 8.5%
Leading product segment(s): Blood biomarker tests (35%) and advanced imaging modalities
Leading material type: Not specified
Leading end use: Hospitals and diagnostic centers (~70%)
Key growth regions: United States (9.4%), Germany (8.9%), Japan (8.6%), China (8.1%), South Korea (7.8%), India (7.5%)
Market Momentum (YoY Path)
The Liver Cancer Diagnostics Market shows a steady and scalable expansion path. Starting at USD 10.6 billion in 2025, the market advances to USD 11.4 billion in 2026 as AI-powered diagnostics and biomarker integration accelerate adoption. By 2028, demand strengthens through wider screening programs and imaging expansion. Growth continues into 2030 and 2031, supported by liquid biopsy adoption and improved accessibility. By 2033, diagnostic standardization and regulatory clarity drive broader clinical usage, culminating in USD 25.8 billion by 2036. The trajectory reflects a transition from innovation-led growth to large-scale population screening adoption.
Why the Market is Growing
The Liver Cancer Diagnostics Market is expanding due to increasing prevalence of hepatitis and liver-related diseases, which elevate screening requirements. AI-powered imaging is significantly reducing interpretation time by 40–60%, improving efficiency across clinical workflows. Additionally, the adoption of liquid biopsy technologies is enabling non-invasive monitoring, making diagnostics more accessible and patient-friendly.
Segment Spotlight
1) Product Type
Blood biomarker tests lead the Liver Cancer Diagnostics Market with a 35% share in 2026. Their dominance is driven by scalability, laboratory integration, and the ability to support multi-parameter analysis for comprehensive cancer screening. Advanced imaging modalities complement this segment by enabling accurate tumor visualization and staging.
2) Material Type
The report emphasizes diagnostic technologies such as biomarker assays, imaging systems, and molecular platforms rather than traditional material classifications. Growth is centered on technological integration, including AI-enabled imaging and standardized biomarker panels that enhance diagnostic accuracy and consistency.
3) End Use
Hospitals and diagnostic centers dominate the Liver Cancer Diagnostics Market, accounting for approximately 70% of demand in 2026. Their leadership reflects the expansion of clinical screening programs, integration of diagnostic protocols, and centralized access to advanced imaging and laboratory infrastructure.
Drivers, Opportunities, Trends, Challenges
Drivers:
Rising hepatitis prevalence and increasing liver cancer incidence are driving demand for early detection solutions. The need for accurate staging and monitoring is reinforcing the adoption of advanced diagnostic technologies.
Opportunities:
AI integration and biomarker discovery optimization present significant opportunities for improving diagnostic accuracy and operational efficiency. Expansion of point-of-care platforms can enhance screening accessibility in underserved regions.
Trends:
The Liver Cancer Diagnostics Market is shifting toward multi-biomarker panels and AI-enhanced imaging. Liquid biopsy adoption is emerging as a key trend, enabling non-invasive and repeatable testing protocols.
Challenges:
Healthcare cost pressures, diagnostic accessibility limitations, and validation hurdles for new technologies may restrict adoption. Rural healthcare infrastructure gaps can delay early detection and screening program effectiveness.
Competitive Landscape
The Liver Cancer Diagnostics Market is characterized by a concentrated structure dominated by established diagnostic and medical device companies with strong regulatory track records. Key players such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, GE Healthcare, Philips Healthcare, Sysmex Corporation, Hitachi Medical Systems, Canon Medical Systems, Bayer AG, and Thermo Fisher Scientific compete based on diagnostic accuracy, cost optimization, and regulatory clearance timelines. Companies with integrated manufacturing capabilities and validated diagnostic platforms hold competitive advantages, while regulatory approvals create distinct market entry barriers.
Get Access of Report Sample: https://www.futuremarketinsights.com/reports/sample/rep-gb-26246
Scope of the Report
Quantitative units: USD 11.4 billion (2026) to USD 25.8 billion (2036), CAGR of 8.5%
Segmentation: By test type, cancer type, and end use
Regions covered: North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa
Countries covered: United States, Germany, France, United Kingdom, Japan, China, South Korea, India, Australia, and 40+ countries
FAQ
How large is the Liver Cancer Diagnostics Market in 2026?
The market is estimated at USD 11.4 billion in 2026.
What will be the market size by 2036?
It is projected to reach USD 25.8 billion by 2036.
What is the expected growth rate?
The market is expected to grow at a CAGR of 8.5% between 2026 and 2036.
Which test type leads the market?
Blood biomarker tests lead with approximately 35% share in 2026.
Who are the primary end users?
Hospitals and diagnostic centers dominate with around 70% share.
Explore More Related Studies Published by FMI Research:
Chaperone-based Therapeutics Market: https://www.futuremarketinsights.com/reports/chaperone-based-therapeutics-market
Substrate Reduction Therapies Market: https://www.futuremarketinsights.com/reports/substrate-reduction-therapies-market
Antineoplastic Combinations Market: https://www.futuremarketinsights.com/reports/antineoplastic-combinations-market
Digital Immunoassays Market: https://www.futuremarketinsights.com/reports/digital-immunoassays-market
Familial Hypercholesterolemia Treatment Market: https://www.futuremarketinsights.com/reports/familial-hypercholesterolemia-treatment-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Why FMI: Decisions that Change Outcomes- https://www.futuremarketinsights.com/why-fmi
Contact Us:
Future Market Insights Inc.
Christiana Corporate, 200 Continental Drive,
Suite 401, Newark, Delaware – 19713, USA
T: +1-347-918-3531
Website: https://www.futuremarketinsights.com
LinkedIn| Twitter| Blogs | YouTube
Have a specific Requirements and Need Assistant on Report Pricing or Limited Budget please contact us - sales@futuremarketinsights.com
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.